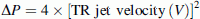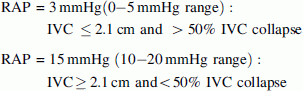- Case Report
- Open access
- Published:
Emergency department diagnosis of atrial and ventricular septal defects, bicuspid aortic valve and pulmonary hypertension
Critical Ultrasound Journal volume 3, pages 35–39 (2011)
Abstract
Introduction
A 41-year-old Cuban man with Down syndrome and mental retardation was brought to the Emergency Department for episodes of worsening shortness of breath over one day. Bedside color Doppler ultrasound subcostal and apical four-chamber examination of the heart revealed atrial and ventricular septal defects with left-to-right shunting of blood. A right ventricular outflow tract view revealed a bicuspid aortic valve, and continuous wave Doppler ultrasound in the right ventricular inflow tract view revealed tricuspid regurgitation and pulmonary hypertension.
Conclusion
Bedside echocardiography with color Doppler and continuous wave Doppler imaging can assist the emergency physician and the critical care physician in the diagnosis of ASD, VSD, bicuspid aortic valve, and pulmonary hypertension in the Down syndrome patient who is short of breath.
Case report
A 41-year-old Cuban man with Down syndrome, and mental retardation was brought to the Emergency Department by his mother, who thought her son looked pale and believed he had episodes of worsening shortness of breath over one day. The patient’s mother reported that he had a congenital heart abnormality but he had never had previous cardiac surgery. He had no known history of deep venous thrombosis or pulmonary embolism. His Emergency Department (ED) vital signs were temperature 98°F, blood pressure 100/70 mmHg, respiratory rate 18 bpm, room air oxygen saturation 100%. Electrocardiogram revealed normal sinus rhythm with a heart rate of 70 bpm and inverted T waves in both the inferior leads and in leads V1–V4. On physical examination he was noted to be a very thin man in minimal respiratory distress. He had a precordial holosystolic murmur. His lungs were clear to auscultation. He had no leg edema or tenderness to palpation. Portable chest X-ray showed no focal consolidation or pleural effusion. CT scan of the chest with and without intravenous contrast revealed no pulmonary embolism, but showed enlargement of the right atrium, right ventricle, and pulmonary arteries. Initial laboratory studies revealed a BNP of 101 pg/mL and a negative troponin I level.
Initial ED bedside echocardiography was performed (see Video Clips S1, S2, S3, S4, and S5, available as supporting information in the online version of this paper). Examination of the heart with a low frequency array cardiac probe in the apical four-chamber view revealed right ventricular hypertrophy and a combined atrial septal defect (ASD) and ventricular septal defect (VSD) with no pericardial effusion (Fig. 1; Video Clip S1). A color Doppler subcostal view revealed a left-to-right shunt from the left ventricle to the right atrium (Fig. 2; Video Clip S2). Right ventricular outflow tract view (RVOT) evaluation of the aortic valve revealed a bicuspid aortic valve (Fig. 3; Video Clip S3). Parasternal short axis view at the level of the papillary muscles showed right ventricular pressure and volume overload with a D-shaped left ventricle present during both systole and diastole and a large dilated right ventricle (Fig. 4; Video Clip S4). Color Doppler evaluation of the tricuspid valve in the right ventricular inflow tract view (RVIT) revealed tricuspid regurgitation (Fig. 5; Video Clip S5). In addition, in the RVIT view (Fig. 6), continuous wave Doppler revealed tricuspid regurgitation with a 2.76 m/s regurgitant jet. Right ventricular systolic pressure (RVSP) was estimated elevated at 45.4 mmHg (greater than 25 mmHg is abnormal) from this tricuspid regurgitant jet velocity using the modified Bernoulli equation and combining this with an estimate of the right atrial pressure (RAP) [1]. Right ventricle systolic pressure (RVSP) = pressure gradient between the RV and RA = 4 × [tricuspid regurgitant Doppler jet velocity (meters/second)]2 + right atrial pressure (RAP) [1]. Right atrial pressure was estimated at 15 mmHg, as the patient had an inferior vena cava (IVC) diameter >2.1 cm and had less than 50% inspiratory collapse of the IVC in the subcostal view of the IVC going into the right atrium (Fig. 7) [1]. Using the modified Bernoulli equation and RAP estimation using IVC inspiratory collapse method recommended by the American Society of Echocardiography [1]:
RVSP = [ΔP (between the RA and RV) + RAP] (valid with no pulmonic stenosis)
-
∆P may be calculated from the tricuspid regurgitant (TR) jet velocity using the modified Bernoulli equation:
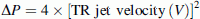
-
RAP may be estimated with subcostal IVC measurement and IVC inspiratory collapse:
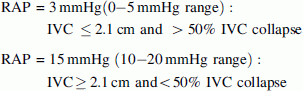
In our patient:

The patient subsequently had a formal echocardiography evaluation performed by the Cardiology department that revealed a large inflow ventricular septal defect, prominent septum primum atrial septal defect, and severe right ventricular hypertrophy, flattening of the intraventricular septum throughout the cardiac cycle, consistent with RV pressure and volume overload. Also shown was a bicuspid aortic valve with fusion of the right and non-coronary cusps. No pericardial effusion was present. Both right and left atria were enlarged. In addition, the formal Cardiology echocardiogram revealed a tricuspid regurgitant jet of 2.8 m/s, with estimated right atrial pressures of 10–15 mmHg suggesting a RVSP of 41–46 mmHg. No pulmonic stenosis was present. The patient was admitted to the hospital for further evaluation by the Cardiology Department.
Discussion
Emergency and critical care physicians should always consider the diagnosis of congenital heart disease in adult patients with Down syndrome with new onset shortness of breath [1–4]. Down syndrome (trisomy 21) is the most common autosomal chromosomal abnormality, affecting 13–22 of every 10,000 live births [5–7]. With life expectancy for these patients having increased from 12 years during the 1940s to 60 years currently, they are more frequently seen in adult emergency departments [8, 9]. The prevalence of congenital heart disease among Down syndrome patients is significant, reported in the literature as 23–58% [10–13]. Atrioventricular septal defects (ASVD, also called endocardial cushion defects), which were found in our patient, are among the most common [7, 10, 11]. The pathogenesis of these defects is unknown, but overexpression of collagen type VI is thought to play a role [9, 14]. Notably, 66% of all patients with ASVD also have Down syndrome, so this is an important association for emergency physicians to recognize [10, 15]. Though our patient was Hispanic, recent research has suggested that Black infants with Down syndrome actually have the highest risk of ASVD, four times that of Hispanic infants, and twice that of Caucasian infants [10].
Once our patient had the diagnosis of pulmonary embolism excluded with a negative CT scan of the chest, bedside ED echocardiography was a useful tool to diagnose his ASD, VSD, bicuspid aortic valve and pulmonary hypertension, which facilitated cardiology evaluation and admission. The majority of adults with significant congenital cardiac disease present to the Emergency Department with symptoms related to cardiac arrhythmias, congestive heart failure, syncope, aortic dissection, endocarditis and pulmonary hypertension [3]. Our patient had shortness of breath, probably related to pulmonary hypertension that resulted from a chronic left-to-right shunt through his ASD and VSD. Our patient had elevated right ventricular pressure and volume overload with a dilated right ventricle with tricuspid regurgitation. Notably, elevated right ventricular systolic pressure may be used to estimate pulmonary artery systolic pressure (as no pulmonic stenosis was present) [1]. Our Emergency Department bedside estimate of RVSP, 45.4 mmHg, was close to the formal Cardiology estimate of RVSP, 41–46 mmHg, reinforcing the potential efficacy of ED bedside echocardiography for cardiac applications. However, fair to moderate interrater reliability (kappa of 0.41) of emergency physician performed versus cardiology performed echocardiography evaluation of IVC inspiratory collapse in non-intubated patients to estimate central venous pressure has been reported [17]. Bedside echocardiography was a useful diagnostic tool to help alter our patient’s management by recognizing the structural cardiac defects and obtaining formal Cardiology consultation. In addition, we limited giving the patient intravenous fluids once we recognized that he had pulmonary hypertension and elevated right atrial pressure with increased intravascular volume.
As many Down syndrome patients presenting to the emergency department, particularly those not accompanied by a parent or caregiver, may not be able to provide a detailed history, thorough physical examination is crucial for swift and appropriate diagnosis and disposition [16]. Bedside ED echocardiography may be a valuable addition to the physical exam. In our patient, a holosystolic murmur on cardiac exam provided a diagnostic clue that we were able to quickly, effectively, and non-invasively investigate further with echocardiography. Rapidly identifying an adult patient with ASD, VSD, bicuspid aortic valve and pulmonary hypertension in the Emergency Department or Intensive Care Unit can expedite the important cardiology and or cardiothoracic surgical consultation that the majority of these patients will require.
Advanced echocardiography evaluation of patients for congenital heart defects and pulmonary hypertension is an operator-dependant modality and emergency and critical care physician screening advanced echocardiography should always be followed by formal Cardiology echocardiography examination.
Conclusion
Bedside echocardiography with color Doppler and continuous wave Doppler imaging can assist the emergency physician and the critical care physician in the diagnosis of ASD, VSD, bicuspid aortic valve and pulmonary hypertension in the Down syndrome patient who is short of breath.
References
Rudski LG, Lai WW, Afilalo J et al (2010) Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 23:685–713
Al-Jarallah AS (2009) Down’s syndrome and the pattern of congenital heart disease in a community with high parental consanguinity. Med Sci Monit 15:CR409–12
Kaemmerer H, Bauer U, Pensl U et al (2008) Management of emergencies in adults with congenital cardiac disease. Am J Cardiol 101:521–525
Calabro R, Limongelli G (2006) Complete atrioventricular canal. Orphanet J Rare Dis 1:8. http://www.OJRD.com/content/1/1/8
Centers for Disease Control and Prevention (CDC) (2006) Improved national prevalence estimates for 18 selected major birth defects—United States, 1999–2001. MMWR Morb Mortal Wkly Rep 54:1301–1305
Collins VR, Muggli EE, Riley M, Palma S, Halliday JL (2008) Is Down syndrome a disappearing birth defect? J Pediatr 152:20–24 (4 e1)
Weijerman ME, van Furth AM, Vonk Noordegraaf A, van Wouwe JP, Broers CJ, Gemke RJ (2008) Prevalence, neonatal characteristics, and first-year mortality of Down syndrome: a national study. J Pediatr 152:15–19
Bittles AH, Bower C, Hussain R, Glasson EJ (2007) The four ages of Down syndrome. Eur J Public Health 17:221–225
Vis JC, Duffels MG, Winter MM et al (2009) Down syndrome: a cardiovascular perspective. J Intellect Disabil Res 53:419–425
Freeman SB, Bean LH, Allen EG et al (2008) Ethnicity, sex, and the incidence of congenital heart defects: a report from the National Down Syndrome Project. Genet Med 10:173–180
Freeman SB, Taft LF, Dooley KJ et al (1998) Population-based study of congenital heart defects in Down syndrome. Am J Med Genet 80:213–217
Kallen B, Mastroiacovo P, Robert E (1996) Major congenital malformations in Down syndrome. Am J Med Genet 65:160–166
Weijerman ME, van Furth AM, van der Mooren MD et al (2010) Prevalence of congenital heart defects and persistent pulmonary hypertension of the neonate with Down syndrome. Eur J Pediatr 169:1195–1199
Gittenberger-de Groot AC, Bartram U, Oosthoek PW et al (2003) Collagen type VI expression during cardiac development and in human fetuses with trisomy 21. Anat Rec A Discov Mol Cell Evol Biol 275:1109–1116
Loffredo CA, Hirata J, Wilson PD, Ferencz C, Lurie IW (2001) Atrioventricular septal defects: possible etiologic differences between complete and partial defects. Teratology 63:87–93
Grossman SA, Richards CF, Anglin D, Hutson HR (2000) Caring for the patient with mental retardation in the emergency department. Ann Emerg Med 35:69–76
Randazzo MR, Snoey ER, Levitt MA, Binder K (2003) Accuracy of emergency physician assessment of left ventricular ejection fraction and central venous pressure using echocardiography. Acad Emerg Med 10:973–977
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MOV 7027 kb)
Supplementary material 2 (MOV 6598 kb)
Supplementary material 3 (MOV 4788 kb)
Supplementary material 4 (MOV 5648 kb)
Supplementary material 5 (MOV 7997 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Riley, D.C., Kim, I., Sacco, D. et al. Emergency department diagnosis of atrial and ventricular septal defects, bicuspid aortic valve and pulmonary hypertension. Crit Ultrasound J 3, 35–39 (2011). https://doi.org/10.1007/s13089-011-0061-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13089-011-0061-8